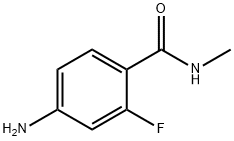
915087-25-1, commonly known as 4-Amino-2-fluoro-N-methylbenzamide, is an important organic compound widely used in pharmaceutical and chemical research. This compound belongs to the benzamide family, which includes molecules that play significant roles in medicinal chemistry and drug development. Due to its unique molecular structure containing amino, fluoro, and amide functional groups, 4-Amino-2-fluoro-N-methylbenzamide has gained attention as a useful intermediate in the synthesis of biologically active compounds.
Researchers and chemical manufacturers frequently use this compound in laboratory studies, particularly in the development of advanced pharmaceutical molecules. Its versatile chemical behavior makes it suitable for various synthetic reactions and research applications. In this article, we explore the chemical properties, synthesis methods, applications, and safety considerations associated with 915087-25-1 (4-Amino-2-fluoro-N-methylbenzamide).
Understanding the fundamental chemical information about this compound helps researchers work with it effectively in laboratory environments.
Chemical Name: 4-Amino-2-fluoro-N-methylbenzamide
CAS Number: 915087-25-1
Molecular Formula: C8H9FN2O
Molecular Weight: Approximately 168.17 g/mol
Chemical Class: Benzamide derivative
Functional Groups: Amino group (-NH2), Fluoro group (-F), Amide group (-CONH)
The presence of multiple functional groups contributes to the compound’s chemical versatility and reactivity. These groups allow the compound to participate in various chemical transformations used in pharmaceutical and organic synthesis.
4-Amino-2-fluoro-N-methylbenzamide features a substituted benzene ring that contains several important functional groups. These groups influence the chemical and physical properties of the molecule.
Key structural elements include:
Benzene Ring Core: Provides aromatic stability.
Amino Group (-NH₂): Enhances reactivity and enables hydrogen bonding.
Fluorine Substitution: Improves chemical stability and influences biological activity.
N-Methyl Amide Group: Contributes to the molecule’s solubility and reactivity.
Fluorine atoms are commonly introduced into pharmaceutical molecules because they can improve metabolic stability and biological activity. This is one reason why compounds like 4-Amino-2-fluoro-N-methylbenzamide are important in medicinal chemistry research.
The physical and chemical characteristics of 915087-25-1 determine how it behaves during storage, handling, and laboratory experiments.
Appearance: Typically a white to off-white crystalline powder
Molecular Weight: 168.17 g/mol
Solubility: Soluble in many organic solvents such as methanol, DMSO, and dimethylformamide
Stability: Stable under standard laboratory conditions when properly stored
Aromatic benzamide compound
Contains reactive amino and amide functional groups
Fluorine atom influences electron distribution within the molecule
Capable of participating in substitution and coupling reactions
These properties make the compound highly useful in research involving synthetic organic chemistry and drug development.
Several synthetic methods can be used to produce 915087-25-1. The choice of synthesis route often depends on the availability of starting materials and the desired purity level.
One of the most common synthesis methods involves forming the amide bond through a reaction between a substituted benzoic acid derivative and methylamine.
Typical steps include:
Starting with a 2-fluoro-4-aminobenzoic acid derivative
Activating the carboxylic acid group using a coupling reagent
Reacting it with methylamine to form the N-methylbenzamide structure
This approach is widely used because it produces good yields and allows precise control of the reaction.
Another synthesis pathway may involve fluorinated aromatic intermediates. Through controlled substitution reactions, the desired amino and amide groups are introduced into the benzene ring.
This approach is useful when preparing fluorinated pharmaceutical intermediates.
Modern organic chemistry often uses catalytic processes to produce complex molecules efficiently. Transition metal catalysts can assist in forming carbon–nitrogen bonds and other important chemical transformations required to synthesize this compound.
Advantages of catalytic synthesis include:
Higher reaction efficiency
Improved selectivity
Scalability for industrial production
One of the primary uses of 4-Amino-2-fluoro-N-methylbenzamide is in pharmaceutical research. Benzamide derivatives are commonly used as building blocks for drug discovery and medicinal chemistry.
Researchers study these compounds because they can interact with biological targets such as enzymes and receptors. The fluorine substitution often enhances biological stability and improves drug-like properties.
Possible areas of pharmaceutical research include:
Anticancer drug development
Anti-inflammatory compound design
Neurological disorder research
Enzyme inhibition studies
Because of its functional groups and chemical structure, this compound serves as an important intermediate in the synthesis of complex therapeutic molecules.
Medicinal chemists frequently modify benzamide derivatives to optimize biological activity. 4-Amino-2-fluoro-N-methylbenzamide offers a flexible structure that can be further modified through various chemical reactions.
Common modifications include:
Substitution on the aromatic ring
Formation of additional amide bonds
Coupling reactions with other organic molecules
Introduction of heterocyclic structures
These modifications help scientists design molecules with improved pharmacological properties such as better absorption, stability, and target specificity.
In addition to pharmaceutical research, 915087-25-1 is also valuable in organic chemistry laboratories. It can be used as a starting material or intermediate in the synthesis of more complex molecules.
Some typical reactions involving this compound include:
Amide coupling reactions
Nucleophilic substitution reactions
Functional group transformations
Formation of heterocyclic compounds
Because of its reactive amino and amide groups, it can participate in numerous organic reactions used in advanced chemical research.
Fluorine-containing organic compounds are widely studied in modern chemistry. Introducing fluorine atoms into molecules often leads to significant improvements in chemical and biological properties.
Benefits of fluorination include:
Increased metabolic stability
Improved binding to biological targets
Enhanced lipophilicity
Better pharmacokinetic properties
The presence of the fluorine atom in 4-Amino-2-fluoro-N-methylbenzamide makes it particularly attractive for drug discovery programs.
As with all research chemicals, proper safety precautions must be followed when handling 915087-25-1 in the laboratory.
Recommended safety measures include:
Wearing protective gloves and safety goggles
Working in a well-ventilated laboratory or fume hood
Avoiding direct contact with skin or eyes
Following laboratory safety guidelines
Researchers should always consult the compound’s Material Safety Data Sheet (MSDS) before working with it.
Proper storage helps maintain the stability and purity of 4-Amino-2-fluoro-N-methylbenzamide.
Recommended storage conditions include:
Store in a cool, dry place
Keep container tightly sealed
Protect from moisture and direct sunlight
Store away from strong oxidizing agents
Following these guidelines ensures the compound remains suitable for research use over time.
The demand for novel pharmaceutical compounds continues to grow, and benzamide derivatives remain an important area of study. 4-Amino-2-fluoro-N-methylbenzamide may play a role in future research developments involving advanced therapeutic agents.
Potential future research directions include:
Development of fluorinated drug candidates
Discovery of new enzyme inhibitors
Design of innovative medicinal chemistry scaffolds
Applications in material science and biotechnology
As research progresses, compounds like this will continue to support innovation in chemical and pharmaceutical sciences.
915087-25-1 (4-Amino-2-fluoro-N-methylbenzamide) is a valuable benzamide derivative widely used in chemical and pharmaceutical research. Its unique combination of amino, fluoro, and amide functional groups makes it a versatile compound for synthetic chemistry and medicinal applications.
From drug discovery and organic synthesis to academic research, this compound plays an important role as a chemical intermediate and research tool. By understanding its chemical properties, synthesis methods, and potential applications, scientists can effectively utilize 4-Amino-2-fluoro-N-methylbenzamide in the development of new technologies and innovative pharmaceutical solutions.