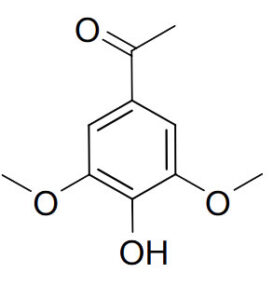
4’-Hydroxy-3’,5’-Dimethoxyacetophenone (CAS No: 2478-38-8) is an important organic compound widely used in pharmaceutical, cosmetic, and chemical industries. Known for its distinctive aromatic structure and biological potential, this compound serves as both a valuable research molecule and a versatile intermediate for complex chemical syntheses.
Chemical Name: 4’-Hydroxy-3’,5’-Dimethoxyacetophenone
CAS Number: 2478-38-8
Molecular Formula: C₁₀H₁₂O₄
Molecular Weight: 196.20 g/mol
Appearance: Off-white to light yellow crystalline powder
Solubility: Soluble in organic solvents such as ethanol, methanol, and chloroform
This compound is a derivative of acetophenone substituted with hydroxyl and methoxy groups, which enhance its stability and reactivity. The presence of phenolic and methoxy functionalities also imparts significant biological and antioxidant properties.
Antioxidant Activity:
The phenolic hydroxyl group contributes to its strong radical-scavenging ability, making it useful in formulations requiring oxidative stability.
Chemical Stability:
4’-Hydroxy-3’,5’-Dimethoxyacetophenone is thermally stable and resistant to light-induced degradation, ensuring long shelf life in chemical and cosmetic applications.
Biological Compatibility:
Its structure supports diverse biological interactions, making it a candidate for pharmacological and biochemical research.
This compound is widely used in drug synthesis and research. It serves as a building block in developing therapeutic agents with anti-inflammatory, antimicrobial, and antioxidant properties. Researchers often explore its derivatives for potential activity against oxidative stress and metabolic disorders.
Owing to its antioxidant nature, 4’-Hydroxy-3’,5’-Dimethoxyacetophenone is used in cosmetic formulations to protect skin cells from free radical damage. It helps in anti-aging, skin-brightening, and UV protection products.
In synthetic chemistry, this compound acts as a precursor for the preparation of natural product analogs and aromatic ethers. It also plays a role in forming advanced materials and fine chemicals through esterification or condensation reactions.
4’-Hydroxy-3’,5’-Dimethoxyacetophenone is frequently studied in biochemical assays due to its phenolic structure, which offers insight into enzyme inhibition, antioxidant potential, and metabolic pathways.
While the compound is relatively safe, standard laboratory precautions should be followed:
Use personal protective equipment (gloves, goggles, lab coat).
Handle in a well-ventilated area.
Store in a cool, dry place, away from direct sunlight and oxidizing agents.
Always refer to the Material Safety Data Sheet (MSDS) for detailed handling instructions.
Excellent antioxidant and protective properties.
Useful as a pharmaceutical and cosmetic intermediate.
Stable and versatile under various chemical conditions.
Valuable for research and development in multiple scientific fields.
4’-Hydroxy-3’,5’-Dimethoxyacetophenone (CAS 2478-38-8) is a multifunctional compound bridging chemistry and biology. Its stability, bioactivity, and structural versatility make it indispensable across pharmaceuticals, cosmetics, and advanced chemical synthesis. As research continues to explore its full potential, this compound stands out as a promising ingredient in both industrial and scientific innovation.