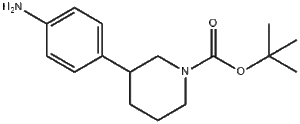
CAS Number: 875798-79-1
Chemical Name: (3-(4-Aminophenyl)piperidine-1-carboxylic acid tert-butyl ester)
IUPAC Name: tert-Butyl 3-(4-aminophenyl)piperidine-1-carboxylate
Synonyms:
3-(4-Aminophenyl)-1-Boc-piperidine
tert-Butyl 3-(4-aminophenyl)piperidine-1-carboxylate
Boc-3-(4-aminophenyl)piperidine
(3-(4-Aminophenyl)piperidin-1-yl) tert-butyl carbonate
Molecular Formula: C₁₆H₂₄N₂O₂
Molecular Weight: ~276.38 g/mol
Appearance: White to off-white crystalline powder
Physical State: Solid
Melting Point: Typically 100–120 °C (may vary depending on purity and source)
Boiling Point: Not applicable (decomposes under high temperature)
Solubility:
Soluble in common organic solvents such as methanol, ethanol, ethyl acetate, dichloromethane (DCM), and DMSO; limited solubility in water
Stability:
Stable under normal laboratory conditions; sensitive to strong acids (Boc group cleavage) and strong oxidizing agents
Functional Groups:
Aromatic amine (–NH₂)
Piperidine ring
tert-Butyl ester (Boc protecting group)
Carbamate linkage (–NH–COO–)
Aromatic benzene ring
Chemical Class:
Protected amine / Carbamate derivative / Piperidine derivative
SMILES (representative):
CC(C)(C)OC(=O)N1CCC(CC1)C2=CC=C(C=C2)N
InChI Key (representative):
(Varies by database; available from chemical registries)
875798-79-1 is commonly used as a protected piperidine building block in pharmaceutical synthesis. The tert-butyl carbamate (Boc) group protects the nitrogen atom during multi-step synthetic processes.
It is particularly valuable for:
Construction of heterocyclic drug candidates
Development of CNS-active molecules
Preparation of kinase inhibitors and receptor-targeting compounds
Intermediate for biologically active compounds
Building block for SAR (Structure–Activity Relationship) studies
Precursor for amine-functionalized derivatives after Boc deprotection
Used in combinatorial chemistry and lead optimization programs
The presence of both an aromatic amine and a protected piperidine makes it highly versatile for further derivatization.
Commonly employed in:
Academic synthetic organic chemistry
Medicinal chemistry research
Drug discovery programs
Multi-step synthesis of advanced intermediates
The Boc protecting group allows selective deprotection under acidic conditions, enabling controlled synthetic transformations.
GHS Classification (typical for similar compounds):
Harmful if swallowed
Causes skin and eye irritation
May cause respiratory irritation
Common Hazard Statements:
H302: Harmful if swallowed
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
Precautionary Statements:
P261: Avoid breathing dust
P280: Wear protective gloves and eye protection
P305 + P351 + P338: IF IN EYES, rinse cautiously with water and remove contact lenses
P301 + P312: IF SWALLOWED, call a doctor if you feel unwell
Handle in a well-ventilated laboratory using appropriate personal protective equipment (PPE).
Typical Purity: 97–99% (Research and Industrial Grade)
Storage Conditions:
Store in a tightly sealed container
Keep in a cool, dry, and well-ventilated area
Protect from moisture and direct sunlight
Avoid strong acids and oxidizing agents
Commercial Use:
Supplied by specialty chemical manufacturers for research and industrial organic synthesis.
Not intended for direct pharmaceutical, food, or diagnostic use without proper regulatory compliance.
875798-79-1 (3-(4-AMINO-PHENYL)-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER) is a versatile protected piperidine derivative widely used as a pharmaceutical intermediate. Its combination of an aromatic amine functionality and Boc-protected nitrogen makes it an essential building block in medicinal chemistry, fine chemical synthesis, and advanced organic research applications.
As a trusted manufacturer and supplier in Hyderabad, SriniChem provides high-quality 875798-79-1 (3-(4-AMINO-PHENYL)-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER) produced under strict quality control standards. Our product, with CAS No: 875798-79-1, is suitable for both research and industrial use. Choose SriniChem for reliable supply, technical expertise, and customer-focused service. Contact SriniChem today for 3-(4-AMINO-PHENYL)-PIPERIDINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER.