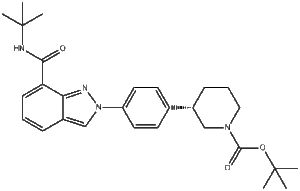
CAS Number: 1476776-84-7
Chemical Name: Niraparib Intermediate 1
IUPAC Name: (Specific IUPAC name may vary depending on supplier classification; available through chemical registries)
Synonyms:
Niraparib Key Intermediate 1;
Niraparib Impurity/Intermediate A;
API Intermediate for Niraparib Synthesis
Molecular Formula: C₁₉H₂₀N₄O (Representative for commonly supplied intermediate; may vary by structural variant)
Molecular Weight: ~320.39 g/mol (Representative value; confirm with supplier COA)
Appearance: Off-white to light yellow crystalline powder
Physical State: Solid
Melting Point: Typically reported between 150–190 °C (may vary depending on purity and salt form)
Boiling Point: Not applicable (organic solid; decomposes before boiling)
Solubility:
Soluble in common organic solvents such as methanol, ethanol, dichloromethane, ethyl acetate, and DMSO; limited solubility in water
Stability:
Stable under recommended storage conditions; sensitive to strong oxidizing agents, excessive heat, and moisture
Functional Groups (Representative):
Heterocyclic aromatic ring systems
Amide or amine functionality
Substituted phenyl ring
Nitrogen-containing fused ring system
Chemical Class:
Heterocyclic organic compound / Pharmaceutical intermediate
SMILES (representative):
(Structure available from chemical registries and supplier databases)
InChI Key (representative):
(Varies by database; available from chemical registries)
1476776-84-7 is widely used as a key intermediate in the synthesis of Niraparib, a PARP (Poly ADP-Ribose Polymerase) inhibitor.
It plays a critical role in:
Construction of the core heterocyclic framework
Introduction of functional groups required for API formation
Final-stage coupling or modification reactions
Used in:
Synthesis of oncology drug candidates
Manufacturing of Niraparib Tosylate (API)
Scale-up production in GMP facilities
Its structural features enable efficient incorporation into the final Niraparib molecule through multi-step synthetic pathways.
Commonly employed in:
Process development and route optimization
Medicinal chemistry research
Impurity profiling studies
Pharmaceutical analytical method development
The compound supports structure–activity relationship (SAR) optimization during anti-cancer drug development.
GHS Classification (Typical for similar pharmaceutical intermediates):
May be harmful if swallowed
Causes skin and eye irritation
May cause respiratory irritation
Common Hazard Statements:
H302: Harmful if swallowed
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
Precautionary Statements:
P261: Avoid breathing dust
P280: Wear protective gloves and eye protection
P305 + P351 + P338: IF IN EYES, rinse cautiously with water and remove contact lenses
P301 + P312: IF SWALLOWED, call a doctor if you feel unwell
Handle in a well-ventilated area using appropriate personal protective equipment (PPE).
Typical Purity: 97–99% (Research and Industrial Grade)
Storage Conditions:
Store in a tightly sealed container
Keep in a cool, dry, and well-ventilated area
Protect from moisture and direct sunlight
Keep away from strong oxidizing agents
Commercial Use:
Available from specialty pharmaceutical intermediate manufacturers for research and industrial API synthesis applications.
Not intended for direct pharmaceutical, food, or diagnostic use without proper regulatory compliance.
1476776-84-7 (Niraparib Intermediate 1) is an important pharmaceutical intermediate used in the synthesis of the PARP inhibitor Niraparib. Its heterocyclic structure and functionalized framework make it a valuable compound in oncology drug manufacturing, medicinal chemistry research, and large-scale API production.
As a trusted manufacturer and supplier in Hyderabad, SriniChem provides high-quality 1476776-84-7 (Niraparib Intermediate 1) produced under strict quality control standards. Our product, with CAS No: 1476776-84-7 , is suitable for both research and industrial use. Choose SriniChem for reliable supply, technical expertise, and customer-focused service. Contact SriniChem today for Niraparib Intermediate 1.