In the field of organic chemistry and industrial chemical manufacturing, organophosphorus compounds play a critical role in the development of advanced materials, pharmaceuticals, and agrochemicals. One such important compound is 122-52-1 (Triethyl Phosphite). Known for its versatile reactivity and usefulness in organic synthesis, triethyl phosphite is widely used as a reagent and intermediate in chemical research and industrial production.
Because of its unique chemical properties and ability to participate in various reactions, this compound has become an essential component in laboratories, pharmaceutical industries, and chemical manufacturing processes. Understanding its chemical profile and research applications helps highlight why it remains valuable in modern chemistry.
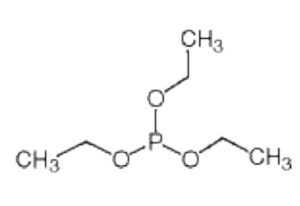
Triethyl phosphite is an organophosphorus compound composed of a phosphorus atom bonded to three ethyl groups through oxygen atoms. It is commonly used as a reagent in organic synthesis and as a precursor for the preparation of other phosphorus-containing compounds.
Chemical Name: Triethyl Phosphite
CAS Number: 122-52-1
Molecular Formula: C6H15O3P
Molecular Weight: 166.16 g/mol
Chemical Class: Organophosphorus compound
Triethyl phosphite is widely recognized for its role as a phosphorus donor and reducing agent in many chemical reactions.
The molecular structure of triethyl phosphite consists of a central phosphorus atom bonded to three ethoxy groups (–OCH2CH3). This structure provides flexibility and reactivity, enabling the compound to participate in multiple organic reactions.
Key structural features include:
A phosphorus atom with three ester bonds
Three ethyl groups connected through oxygen atoms
A stable yet reactive organophosphorus framework
These structural characteristics allow triethyl phosphite to function effectively as a reagent in various synthetic processes.
Triethyl phosphite possesses several properties that make it suitable for laboratory and industrial use.
Appearance: Colorless to pale yellow liquid
Odor: Mild characteristic odor
Boiling Point: Approximately 156°C
Density: Around 0.96 g/cm³
Solubility: Soluble in many organic solvents such as ethanol, ether, and benzene
These properties make it easy to handle in controlled chemical environments and suitable for use in synthesis and manufacturing.
One of the most significant uses of 122-52-1 (Triethyl Phosphite) is in organic synthesis. Chemists use it as a reagent to introduce phosphorus-containing functional groups into organic molecules.
Some common reactions involving triethyl phosphite include:
Triethyl phosphite participates in the Michaelis–Arbuzov reaction, where it reacts with alkyl halides to form phosphonates. These compounds are important intermediates in organic chemistry and pharmaceutical synthesis.
The compound can act as a mild reducing agent in certain organic transformations.
Triethyl phosphite can also serve as a ligand in coordination chemistry, forming complexes with transition metals used in catalytic reactions.
In pharmaceutical research, triethyl phosphite is used as a starting material or intermediate for the synthesis of biologically active compounds.
Researchers utilize it for:
Synthesizing pharmaceutical intermediates
Developing new drug molecules
Preparing phosphorus-based medicinal compounds
Supporting catalytic reactions used in drug synthesis
Its ability to introduce phosphorus groups into molecules makes it particularly valuable in medicinal chemistry.
Beyond research laboratories, triethyl phosphite has numerous industrial applications due to its chemical versatility.
1. Agrochemical Production
Triethyl phosphite is used in the synthesis of pesticide and herbicide intermediates.
2. Plastic Additives
It can be used in the preparation of stabilizers and additives for polymer materials.
3. Flame Retardant Manufacturing
Organophosphorus compounds derived from triethyl phosphite are often used in flame-retardant formulations.
4. Chemical Intermediates
It serves as an intermediate for the production of phosphonates and phosphates used in various industrial processes.
Researchers in organic chemistry and material science rely on 122-52-1 (Triethyl Phosphite) for exploring new chemical reactions and developing innovative materials.
Its importance in research stems from:
Versatility in reaction mechanisms
Compatibility with many organic compounds
Role in forming phosphorus-containing molecules
Ability to participate in catalytic systems
Because of these advantages, the compound continues to be widely studied in academic and industrial laboratories.
Like many chemical reagents, triethyl phosphite should be handled with proper safety precautions.
Recommended safety practices include:
Using gloves, safety goggles, and protective clothing
Working in well-ventilated laboratory areas or fume hoods
Storing the compound in tightly sealed containers away from heat sources
Following established chemical safety guidelines
Proper storage and handling help maintain safety and prevent chemical degradation.
As industries continue to develop new materials and pharmaceuticals, the demand for versatile reagents like triethyl phosphite is expected to grow.
Future research may focus on:
Developing advanced catalytic reactions
Designing new phosphorus-based pharmaceutical compounds
Creating innovative flame-retardant and polymer materials
Improving environmentally friendly chemical synthesis methods
These developments will further strengthen the role of triethyl phosphite in chemical innovation.
122-52-1 (Triethyl Phosphite) is a valuable organophosphorus compound widely used in chemical synthesis, pharmaceutical research, and industrial manufacturing. Its unique structure, versatile reactivity, and ability to participate in multiple chemical reactions make it an essential reagent for chemists and researchers.
From organic synthesis and agrochemical production to pharmaceutical development, triethyl phosphite continues to play a crucial role in advancing chemical science and industrial technology.