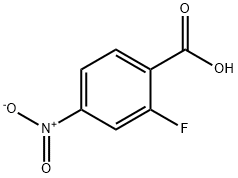
403-24-7 (2-Fluoro-4-nitrobenzoic acid) is an important fluorinated aromatic compound widely used in chemical and pharmaceutical research. This compound belongs to the class of substituted benzoic acids and contains both a fluorine atom and a nitro group attached to the benzene ring. The presence of these functional groups significantly influences its chemical reactivity, making it a valuable intermediate in organic synthesis and industrial chemical processes.
Fluorinated organic compounds have gained considerable attention in modern chemistry due to their unique physical, chemical, and biological properties. In particular, 2-Fluoro-4-nitrobenzoic acid plays an important role in pharmaceutical development, agrochemical synthesis, and advanced materials research. Its versatile structure allows chemists to perform various chemical transformations that lead to the creation of complex molecules.
This article explores the chemical properties, synthesis methods, applications, and safety considerations of 403-24-7 (2-Fluoro-4-nitrobenzoic acid) in detail.
Understanding the fundamental chemical information about 2-Fluoro-4-nitrobenzoic acid helps researchers handle and utilize the compound effectively.
Chemical Name: 2-Fluoro-4-nitrobenzoic acid
CAS Number: 403-24-7
Molecular Formula: C7H4FNO4
Molecular Weight: Approximately 185.11 g/mol
Chemical Class: Fluorinated aromatic carboxylic acid
Functional Groups: Carboxylic acid (-COOH), nitro group (-NO₂), and fluorine substitution
The combination of these functional groups gives the compound unique electronic and structural characteristics that are beneficial in many chemical reactions.
2-Fluoro-4-nitrobenzoic acid consists of a benzene ring substituted with three key functional groups:
Fluorine atom (F) at the second position
Nitro group (-NO₂) at the fourth position
Carboxylic acid group (-COOH) attached to the aromatic ring
These substituents strongly influence the electronic distribution of the aromatic ring. The nitro group is a strong electron-withdrawing group, while fluorine also affects the electron density due to its electronegativity. As a result, the compound exhibits unique chemical reactivity and stability.
The molecular structure makes it particularly useful as an intermediate for synthesizing more complex organic molecules used in pharmaceuticals and agrochemicals.
Understanding the physical and chemical properties of 403-24-7 helps researchers determine how the compound behaves in different laboratory and industrial conditions.
Appearance: White to light yellow crystalline powder
Molecular Weight: 185.11 g/mol
Melting Point: Typically within a moderate temperature range depending on purity
Solubility: Soluble in polar organic solvents such as methanol, ethanol, and dimethyl sulfoxide
Stability: Stable under standard storage conditions
Aromatic carboxylic acid compound
Strong electron-withdrawing nitro group
Fluorine substitution influences reactivity
Suitable for substitution and coupling reactions
These chemical characteristics make 2-Fluoro-4-nitrobenzoic acid a versatile compound for synthetic chemistry.
Several synthetic approaches can be used to produce 403-24-7 (2-Fluoro-4-nitrobenzoic acid). The method chosen often depends on the availability of starting materials and desired production scale.
One of the most common methods involves nitration of fluorobenzoic acid derivatives. In this reaction, a nitrating mixture introduces a nitro group onto the aromatic ring at the desired position.
Typical reagents used include:
Nitric acid
Sulfuric acid as a catalyst
By carefully controlling reaction conditions such as temperature and concentration, the nitro group can be selectively introduced to form 2-Fluoro-4-nitrobenzoic acid.
Another possible synthesis route involves introducing a fluorine atom into a nitrobenzoic acid derivative. This process may involve specialized fluorination reagents and controlled reaction conditions to ensure selective substitution.
Fluorination reactions are important in modern chemistry because fluorine atoms can significantly influence molecular properties.
In some cases, chemists may use multi-step synthesis methods to construct the molecule from simpler starting materials. These methods often include:
Aromatic substitution reactions
Oxidation reactions
Functional group transformations
This approach allows for better control over product purity and yield.
2-Fluoro-4-nitrobenzoic acid is widely used across multiple industries due to its chemical versatility.
One of the most significant uses of this compound is as a pharmaceutical intermediate. Many drug molecules contain fluorinated aromatic structures because they improve drug stability and biological activity.
Researchers use 2-Fluoro-4-nitrobenzoic acid in the synthesis of compounds that may be investigated for:
Anti-inflammatory activity
Antibacterial properties
Anticancer research
Enzyme inhibition studies
The presence of fluorine often enhances the effectiveness of pharmaceutical molecules.
Fluorinated aromatic compounds are also important in agricultural chemistry. 2-Fluoro-4-nitrobenzoic acid can be used as a precursor for synthesizing agrochemical products such as:
Herbicides
Pesticides
Crop protection agents
These chemicals help improve agricultural productivity and protect crops from pests and diseases.
Organic chemists frequently use 2-Fluoro-4-nitrobenzoic acid as a building block in synthetic reactions. It can undergo several transformations including:
Reduction reactions
Amide formation
Esterification
Nucleophilic aromatic substitution
These reactions enable chemists to create complex molecules used in research and industrial applications.
Universities and research institutions use this compound to study reaction mechanisms, fluorinated compounds, and aromatic substitution chemistry. It serves as a useful model compound for studying the behavior of electron-withdrawing substituents in aromatic systems.
Fluorinated compounds are highly valuable in modern chemistry due to their unique properties. Introducing fluorine atoms into organic molecules can significantly affect their behavior.
Key advantages include:
Improved chemical stability
Enhanced biological activity
Increased resistance to metabolic degradation
Better interaction with biological targets
Because of these benefits, fluorinated compounds like 2-Fluoro-4-nitrobenzoic acid are widely used in drug discovery and advanced chemical research.
Like many laboratory chemicals, 403-24-7 (2-Fluoro-4-nitrobenzoic acid) must be handled carefully to ensure safety.
Recommended precautions include:
Wearing protective gloves and safety goggles
Working in a well-ventilated laboratory environment
Avoiding inhalation of dust or vapors
Preventing direct contact with skin and eyes
Researchers should always consult the Material Safety Data Sheet (MSDS) before using the compound.
Proper storage is essential to maintain the quality and stability of the compound.
Recommended storage conditions include:
Store in a cool and dry place
Keep the container tightly sealed
Protect from moisture and direct sunlight
Store away from strong oxidizing agents
Following these guidelines ensures long-term stability for research or industrial use.
As the demand for fluorinated compounds continues to grow, 2-Fluoro-4-nitrobenzoic acid is expected to remain an important intermediate in chemical research and industrial production.
Future research may focus on:
Development of new fluorinated pharmaceuticals
Advanced synthetic methodologies
Novel agrochemical formulations
Applications in materials science
These research directions highlight the ongoing importance of this compound in modern chemistry.
403-24-7 (2-Fluoro-4-nitrobenzoic acid) is a valuable fluorinated aromatic compound widely used in pharmaceutical research, agrochemical development, and organic synthesis. Its unique molecular structure, containing fluorine, nitro, and carboxylic acid functional groups, provides significant chemical versatility.