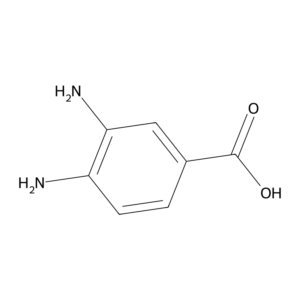
In the field of organic and medicinal chemistry, certain aromatic compounds stand out for their structural importance and versatile applications. One such compound is 3,4-Diaminobenzoic Acid (CAS No: 619-05-6). Known for its amino-substituted benzoic acid structure, this compound serves as a valuable intermediate in pharmaceutical research, dye production, and organic synthesis.
3,4-Diaminobenzoic Acid is a benzoic acid derivative containing two amino groups (-NH₂) positioned at the 3rd and 4th carbons of the benzene ring, along with a carboxylic acid group (-COOH). This arrangement makes it a key building block for synthesizing more complex organic molecules.
IUPAC Name: 3,4-Diaminobenzoic Acid
Molecular Formula: C₇H₈N₂O₂
Molecular Weight: 152.15 g/mol
CAS Number: 619-05-6
Appearance: Generally appears as an off-white to light brown crystalline solid
Melting Point: ~ 256 °C (with decomposition)
Solubility: Moderately soluble in water; soluble in polar solvents
Functional Groups: Carboxylic acid (-COOH), aromatic amines (-NH₂)
Reactivity: Reactive towards acylation, diazotization, and condensation reactions
Used as an intermediate in the synthesis of drug molecules.
Plays a role in developing antibacterial, antifungal, and anticancer compounds.
Structural resemblance to p-aminobenzoic acid (PABA) allows exploration in vitamin-related chemistry.
Serves as a precursor for azo dye synthesis.
Amino groups enable coupling reactions with diazonium salts, producing vibrant pigments.
Useful in designing heterocyclic compounds.
Acts as a raw material in preparing polymers, coatings, and specialty chemicals.
Explored in enzyme inhibition and biochemical assays due to its reactive amino groups.
Like many aromatic amines, 3,4-Diaminobenzoic Acid requires careful handling:
Avoid inhalation and direct skin contact.
Use protective gloves, goggles, and laboratory coats.
Store in a cool, dry, well-ventilated place.
Dispose of waste material following hazardous chemical guidelines.
The dual functionality of amino and carboxylic acid groups in this compound makes it highly versatile. Its role as an intermediate across pharmaceuticals, dyes, and advanced organic synthesis underlines its industrial and research significance.
3,4-Diaminobenzoic Acid (CAS No: 619-05-6) is more than just a chemical intermediate—it’s a critical compound in modern chemistry, bridging research and industrial applications. With its multifunctional structure, it continues to play a vital role in developing new medicines, dyes, and innovative chemical products.