CAS Number: 24065-33-6
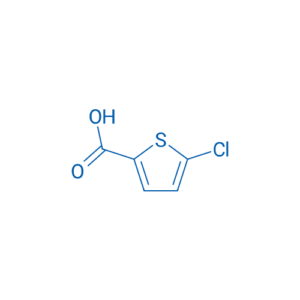
Chemical Name: 5-Chlorothiophene-2-carboxylic acid
IUPAC Name: 5-Chlorothiophene-2-carboxylic acid
Synonyms:
5-Chloro-2-thiophenecarboxylic acid;
5-Chloro-2-carboxythiophene;
5-Chlorothiophene-2-carboxylate (related forms)
Molecular Formula: C₅H₃ClO₂S
Molecular Weight: ~162.59 g/mol
Appearance: White to off-white crystalline powder
Physical State: Solid
Melting Point: Typically reported in the range of 150–156 °C (may vary depending on purity)
Boiling Point: Not applicable (decomposes before boiling under normal pressure)
Solubility:
Soluble in common organic solvents such as methanol, ethanol, acetone, ethyl acetate, and DMSO; limited solubility in water
Stability:
Stable under standard laboratory conditions; sensitive to strong oxidizing agents and strong bases
Functional Groups:
Carboxylic acid group (–COOH)
Chlorine substituent (–Cl)
Heteroaromatic thiophene ring (sulfur-containing five-membered ring)
Chemical Class:
Halogenated thiophene derivative / Heterocyclic carboxylic acid
SMILES (representative):
O=C(O)C1=CC(Cl)=CS1
InChI Key (representative):
(Varies by database; available from chemical registries)
24065-33-6 is widely used as an important building block in organic synthesis, particularly for:
Synthesis of pharmaceutical intermediates
Development of agrochemical compounds
Preparation of biologically active heterocyclic molecules
The thiophene ring system is frequently incorporated into drug molecules due to its aromatic stability and bioisosteric properties.
Used in:
Cross-coupling reactions (e.g., Suzuki, Heck reactions after suitable activation)
Formation of amides, esters, and acid derivatives
Construction of substituted thiophene frameworks
Its chloro substituent enables further functionalization through substitution or metal-catalyzed coupling reactions.
Commonly employed in:
Medicinal chemistry research
Heterocyclic compound development
Structure–activity relationship (SAR) studies
Material science research involving sulfur-containing aromatic systems
The presence of both a reactive carboxylic acid group and a halogen substituent provides versatility for multi-step synthesis.
GHS Classification (typical for similar compounds):
Harmful if swallowed
Causes skin irritation
Causes serious eye irritation
May cause respiratory irritation
Common Hazard Statements:
H302: Harmful if swallowed
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
Precautionary Statements:
P261: Avoid breathing dust
P280: Wear protective gloves and eye protection
P305 + P351 + P338: IF IN EYES, rinse cautiously with water and remove contact lenses
P301 + P312: IF SWALLOWED, call a doctor if you feel unwell
Handle in a well-ventilated area using appropriate personal protective equipment (PPE).
Typical Purity: 97–99% (Research and Industrial Grade)
Storage Conditions:
Store in a tightly sealed container
Keep in a cool, dry, and well-ventilated area
Protect from moisture and direct sunlight
Keep away from strong oxidizing agents and bases
Commercial Use:
Available from specialty chemical manufacturers for research and industrial organic synthesis applications.
Not intended for direct pharmaceutical, food, or diagnostic use without proper regulatory compliance.
24065-33-6 (5-Chlorothiophene-2-carboxylic acid) is an important halogenated thiophene derivative widely utilized as a versatile intermediate in pharmaceutical, agrochemical, and fine chemical synthesis. Its combination of a reactive carboxylic acid group and chloro-substituted heteroaromatic ring makes it a valuable building block for advanced organic synthesis and research applications.
As a trusted manufacturer and supplier in Hyderabad, SriniChem provides high-quality 24065-33-6 (5-Chlorothiophene-2-carboxylic acid) produced under strict quality control standards. Our product, with CAS No: 24065-33-6, is suitable for both research and industrial use. Choose SriniChem for reliable supply, technical expertise, and customer-focused service. Contact SriniChem today for 5-Chlorothiophene-2-carboxylic acid.