In the world of organic chemistry and pharmaceutical innovation, specialized chemical intermediates play a crucial role in developing advanced compounds and materials. One such important compound is 2362-12-1 (4-Bromo-2-methylphenol). This brominated phenolic compound has gained attention among researchers and chemical manufacturers due to its versatile reactivity and usefulness in organic synthesis.
With its unique structural features and functional properties, 4-Bromo-2-methylphenol serves as an essential intermediate in chemical research, pharmaceutical development, and industrial manufacturing. Understanding its chemical profile and applications helps explain why it remains valuable in modern scientific studies.
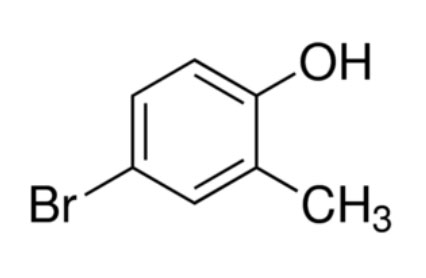
4-Bromo-2-methylphenol is an aromatic compound belonging to the phenol family. It contains a bromine atom and a methyl group attached to a phenolic benzene ring, giving it distinctive chemical behavior.
Chemical Name: 4-Bromo-2-methylphenol
CAS Number: 2362-12-1
Molecular Formula: C7H7BrO
Molecular Weight: Approximately 187.04 g/mol
Chemical Class: Brominated phenolic compound
Phenolic compounds like this are widely studied because they serve as building blocks in many chemical reactions and industrial processes.
The structure of 4-Bromo-2-methylphenol consists of a benzene ring substituted with three functional groups:
A hydroxyl group (-OH) that defines the phenol structure
A bromine atom (Br) positioned at the fourth carbon
A methyl group (-CH₃) located at the second carbon
These substituents influence the compound’s reactivity and allow it to participate in a wide range of chemical reactions.
Aromatic phenolic framework
Halogen substitution that enhances reactivity
Functional groups suitable for chemical modification
Because of these characteristics, the compound is frequently used in the synthesis of more complex molecules.
The physical and chemical properties of 2362-12-1 (4-Bromo-2-methylphenol) make it suitable for laboratory research and industrial applications.
Appearance: White to light-colored crystalline solid
Melting Point: Typically within a moderate temperature range
Solubility: Soluble in many organic solvents such as ethanol and ether
Reactivity: Active in electrophilic substitution and coupling reactions
These properties allow the compound to be easily handled and incorporated into various chemical processes.
One of the primary roles of 4-Bromo-2-methylphenol is as a chemical intermediate in organic synthesis. Chemists often use brominated phenols because the bromine atom can participate in substitution and coupling reactions.
1. Cross-Coupling Reactions
The bromine atom allows the compound to participate in coupling reactions such as Suzuki or Heck reactions, which are widely used in modern organic synthesis.
2. Functional Group Transformations
The phenolic hydroxyl group can undergo modifications, enabling the production of derivatives with different chemical properties.
3. Aromatic Compound Development
Researchers use this compound as a precursor to produce other aromatic molecules for pharmaceutical and industrial purposes.
Phenolic compounds play a significant role in medicinal chemistry. 4-Bromo-2-methylphenol serves as a useful intermediate for synthesizing molecules that may have potential biological activity.
In pharmaceutical research, it can be used for:
Development of pharmaceutical intermediates
Synthesis of biologically active molecules
Research into antimicrobial and antioxidant compounds
Structural modification of drug candidates
The presence of bromine and phenolic functional groups makes it easier to design molecules with specific biological properties.
Beyond academic research, 2362-12-1 (4-Bromo-2-methylphenol) also contributes to several industrial chemical processes.
Chemical Manufacturing
It serves as an intermediate for producing specialty chemicals and advanced organic compounds.
Material Science
Researchers may use brominated phenolic compounds in the development of polymers and specialty materials.
Agrochemical Production
Some derivatives of phenolic compounds are used in the production of agricultural chemicals and protective agents.
These applications highlight the compound’s importance in industrial chemical development.
Modern chemical research focuses heavily on discovering new compounds with improved performance, biological activity, or industrial efficiency. 4-Bromo-2-methylphenol contributes to this process by acting as a flexible and reactive building block.
Researchers value the compound because it:
Enables efficient molecular modification
Supports the synthesis of complex organic structures
Provides a foundation for exploring new chemical reactions
Assists in the development of innovative pharmaceutical and industrial compounds
Its structural versatility makes it an important component in many ongoing research projects.
As with many laboratory chemicals, 4-Bromo-2-methylphenol should be handled with appropriate safety precautions.
Recommended safety measures include:
Wearing gloves, protective clothing, and safety goggles
Working in well-ventilated laboratory environments
Avoiding direct contact with skin or eyes
Storing the compound in sealed containers away from moisture and heat
Following proper safety procedures helps maintain safe laboratory operations.
The demand for specialized organic intermediates continues to grow as industries seek new materials, pharmaceuticals, and chemical technologies. Compounds like 2362-12-1 (4-Bromo-2-methylphenol) will likely remain important in these developments.
Future research may explore:
New pharmaceutical derivatives
Advanced polymer materials
Environmentally friendly chemical synthesis methods
Innovative catalytic reactions involving brominated phenols
These advancements will further expand the compound’s applications in science and industry.
2362-12-1 (4-Bromo-2-methylphenol) is a valuable aromatic compound widely used in modern chemical research and development. Its unique structure, which combines phenolic and brominated functionalities, makes it a versatile intermediate for organic synthesis, pharmaceutical research, and industrial chemical production.
As scientific innovation continues to expand, compounds like 4-Bromo-2-methylphenol will remain essential tools for researchers and manufacturers seeking to develop new chemical technologies and advanced materials.