CAS Number: 133627-45-9
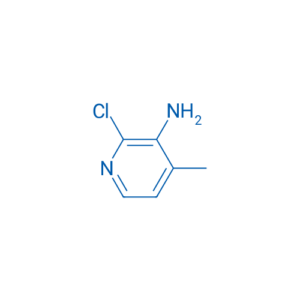
Chemical Name: 3-Amino-2-chloro-4-methylpyridine
IUPAC Name: 3-Amino-2-chloro-4-methylpyridine
Synonyms:
2-Chloro-4-methyl-3-aminopyridine
4-Methyl-2-chloro-3-aminopyridine
Molecular Formula: C₆H₇ClN₂
Molecular Weight: ~142.59 g/mol
133627-45-9 is a substituted pyridine derivative containing multiple functional groups that contribute to its synthetic versatility.
Pyridine ring (heteroaromatic nitrogen-containing ring)
Amino group (–NH₂) at the 3-position
Chloro substituent (–Cl) at the 2-position
Methyl group (–CH₃) at the 4-position
Primary amine
Aryl chloride
Heteroaromatic nitrogen
Alkyl substituent
Halogenated aminopyridine
Substituted heteroaromatic compound
Pharmaceutical intermediate
Appearance: Light yellow to beige crystalline solid
Physical State: Solid
Melting Point: Typically 80–120 °C (may vary by purity and supplier)
Solubility:
Soluble in organic solvents such as methanol, ethanol, acetone, and dichloromethane
Slightly soluble in water
Forms salts with acids due to the amino group
Stability:
Stable under normal laboratory conditions; sensitive to strong oxidizing agents
The compound exhibits dual reactivity due to the presence of both amino and chloro substituents.
Amino Group (–NH₂):
Undergoes acylation
Participates in Schiff base formation
Forms amides, ureas, and heterocycles
Chloro Group (–Cl):
Suitable for nucleophilic aromatic substitution (under appropriate conditions)
Useful in palladium-catalyzed cross-coupling reactions
Pyridine Nitrogen:
Can coordinate with metals
Forms salts with acids
3-Amino-2-chloro-4-methylpyridine is widely used in medicinal chemistry as a building block for:
Antimicrobial agents
Anti-inflammatory compounds
CNS-active molecules
Kinase inhibitors
Nitrogen-containing drug candidates
Substituted pyridines are commonly found in approved pharmaceutical molecules due to their favorable pharmacokinetic properties.
This compound plays a role in the synthesis of:
Crop protection intermediates
Herbicide building blocks
Fungicide research compounds
The pyridine scaffold is widely utilized in modern agrochemical formulations.
Used in:
Heterocyclic compound synthesis
Ligand and catalyst development
Structure–activity relationship (SAR) studies
Advanced academic research
Its multifunctional nature makes it suitable for complex multi-step synthetic routes.
Substituted aminopyridines like 133627-45-9 are highly valued because they:
Provide multiple functionalization options
Enable cross-coupling chemistry
Serve as stable heterocyclic frameworks
Allow structural diversification in drug discovery
The presence of both electron-donating (amino) and electron-withdrawing (chloro) groups creates a balanced electronic system useful for controlled chemical transformations.
May cause skin and eye irritation
Harmful if swallowed
May cause respiratory irritation
Use gloves and protective eyewear
Work in a well-ventilated laboratory or fume hood
Avoid inhalation of dust
Keep container tightly sealed
Store in a cool, dry place
Protect from moisture and light
Keep away from strong oxidizing agents
Typical Commercial Purity: 97–99% (Research and Industrial Grade)
133627-45-9 (3-Amino-2-chloro-4-methylpyridine) is a versatile heteroaromatic intermediate widely used in pharmaceutical, agrochemical, and fine chemical synthesis. Its combination of amino, chloro, and methyl substituents on a pyridine ring makes it highly valuable for medicinal chemistry and advanced organic synthesis.
As a trusted manufacturer and supplier in Hyderabad, SriniChem provides high-quality 133627-45-9 (3-Amino-2-chloro-4-methylpyridine) produced under strict quality control standards. Our product, with CAS No: 133627-45-9, is suitable for both research and industrial use. Choose SriniChem for reliable supply, technical expertise, and customer-focused service. Contact SriniChem today for 3-Amino-2-chloro-4-methylpyridine.